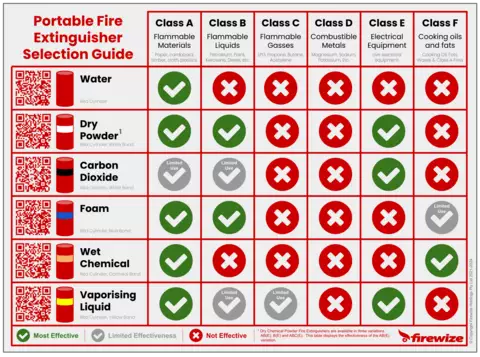
- Fire Classification
Fire classification is a system or method of categorising a fire according to the type(s) of fuel and if live electrical equipment is present. There are six classes of fire;
- Class A — Carbonaceous Solids;
- Class B — Flammable and Combustible Liquids;
- Class C — Flammable Gases
- Class D — Combustible Metals
- Class E — Energized Electrical Equipment
- Class F — Cooking Oils, Fats & Waxes
The performance (ability to control a Class A or Class B fire) of a portable fire extinguisher is denoted by a numerical prefix, and can be found printed on the side of all portable fire extinguishers.
Class A — Carbonaceous Solids
A carbonaceous solid is one which contains the chemical element carbon as the basic fuel. In the built environment, a carbonaceous solid is the most common type of fuel, and includes elements such as paper, cardboard, timber, natural fibres & materials, rubber, plastics, grass, garden clippings and coal.
Class B — Flammable and Combustible Liquids
A flammable liquid is a liquid, or mixtures of liquids, or liquids containing solids in solution or suspension (for example, paints, varnishes, lacquers, etc.) which; (1) give off a flammable vapour at temperatures of not more than 60 °C, closed-cup test, or not more than 65.6 °C, open-cup test, normally referred to as the flash point; or (2) has a flash point of not more than 93°C2 .
A combustible liquid according to the globally harmonised system for the classification and labelling of chemicals (GHS) is any liquid, other than a flammable liquid, that has a flash point, and a fire point less than its boiling point.
Class C — Flammable Gases
Gases can be classified into three groups: oxidisers, inert gases and flammable gases.
- Oxidisers, such as oxygen and chlorine, are not flammable on their own but will act as an oxidant and aid combustion.
- Inert gases are not combustible at all, and are sometimes used in fire suppression systems. Carbon dioxide and helium are examples of inert gases.
- Flammable gases can be explosive when mixed with air in the right proportions. Hydrogen, butane, methane and ethylene are examples of flammable gases.
Class D — Combustible Metals
A combustible metal is any elemental metal or metal alloy that will burn.
In the periodic table of elements there are six metals in Group IA are called alkali metals and they are among the most chemically reactive elements known. In their pure forms, the alkali metals (lithium, sodium, potassium, rubidium, and cesium) are soft, shiny metals with low melting points. Alkali metals react readily with air and moisture.
Class E — Energised Electrical Equipment
Energised electrical equipment includes any equipment that is either energised or has the potential to be energised that could pose a risk of fire. Common energised electrical equipment includes high voltage cables, electrical switchboards, mechanical switchboards and the like. The risk from energised electrical equipment include the dange from electrocution from an extinguishing agent such as water.
Class F — Cooking Oils, Fats & Waxes
Cooking oils, fats and waxes are a range of compounds that when they reach their autoignition temperature can spontaneously combust.